Rochelle Thuynsma, Head of Products: Technical
Introduction
Soil probiotics and prebiotics improve soil function through different mechanisms. Probiotics are live microbial inoculants (e.g., Bacillus, Pseudomonas, Azospirillum, Rhizobium, Trichoderma) that must establish in the rhizosphere to deliver benefits such as nutrient solubilisation, hormone production and pathogen suppression. Their performance is often inconsistent because establishment depends on environmental compatibility (Bashan et al., 2014; Kaminsky et al., 2019).
Prebiotics, by contrast, are non-living substrates or bioactive compounds that stimulate indigenous microbes. Materials such as carbohydrates, organic acids, amino compounds, humic substances and seaweed extracts enhance microbial biomass and activity, improving nutrient mineralisation, phosphorus mobilisation and rhizosphere turnover in a more stable and predictable manner (Canarini et al., 2019; Liu et al., 2020).
RELEASE LPH™ mode of action
RELEASE LPH™ (M441) functions as a soil prebiotic. It contains seaweed extracts (Ascophyllum nodosum, Durvillaea potatorum, Ecklonia maxima) plus organic and amino acids that stimulate microbial growth, nutrient release and root development.
Organic acids enhance nutrient availability via chelation, improved solubility and redox-mediated microbial metabolism (Nardi et al., 2002). Seaweed-derived carbon fractions, polysaccharides and amino compounds act as readily metabolised substrates that increase microbial biomass, enzyme activity, nitrogen turnover, phosphorus solubilisation and micronutrient mobilisation.
Seaweed extracts also directly stimulate plant physiology by promoting meristem activity, lateral roots and root hairs, expanding absorptive surface area and rhizodeposition. This strengthens plant–microbe feedback loops that further enhance nutrient availability.
Greater root biomass increases cytokinin synthesis and transport to shoots, supporting canopy growth, delayed senescence, higher photosynthetic capacity and improved reproductive retention, ultimately contributing to higher yield potential (Sakakibara, 2006).
Tried and tested
Field trials conducted with RELEASE LPH™ across multiple perennial cropping systems demonstrate that these biological and physiological mechanisms translate into measurable agronomic responses.
Root system expansion was evident in both grapevine and citrus production systems, where percentage root coverage increased from 13.5% to 20% in table grape ‘Sweet Globe’ grown in clay soil, and from 10.9% to 15.7% in lemon ‘Fino’ grown in loam-silt soil (Figure 1). These increases represent approximately 45-50% greater soil exploration capacity, directly supporting improved nutrient and water uptake.
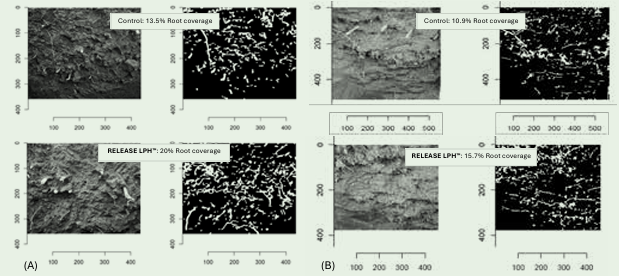
Figure 1: Percentage (%) root coverage after RELEASE LHP™ application on (A) Table Grapes “Sweet Globe” in clay soil, and (B) Lemon “Fino” in loam-silt, 15 L/ha applied four times (60 L per season).
Stimulation of the rhizosphere microbiome accompanied these structural changes. Soil microbial biomass carbon increased significantly following application, together with an elevated fungi: bacteria ratio in both grape and hazelnut systems (Figure 2). Such shifts indicate enhanced biological activity, greater enzymatic turnover, and improved nutrient cycling efficiency within the soil matrix.
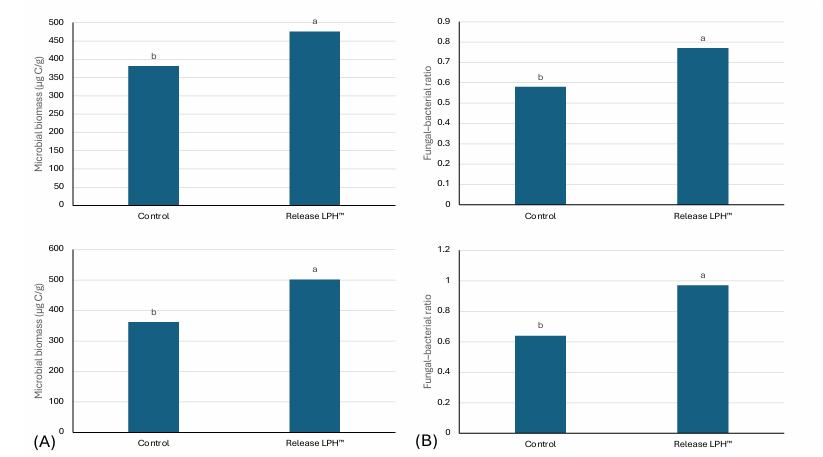
Figure 2: Microbial and fungi:bacteria ratio change after RELEASE LPH™ application on (A) Table Grapes ‘Sweet Globe’ in clay soil, and (B) Hazelnuts ‘TDG’ in clay loam soil, 15 L/ha applied four times (60 L per season). Treatment means differed significantly based on Duncan’s Multiple Range Test at the 5% significance level (p ≤ 0.05).
Improved soil biological function and root architecture were reflected in plant physiological performance. In cherry ‘Regina’, treated trees exhibited greater root biomass and increased stomatal conductance compared with untreated controls (Figure 3), indicating enhanced hydraulic conductivity and photosynthetic capacity.
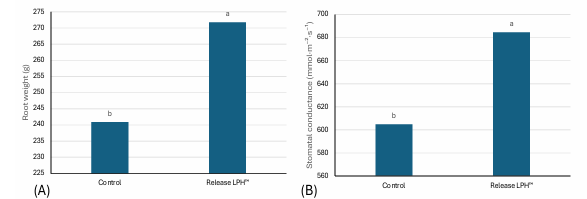
Figure 3: Physiological effects after Release LPH™ application on (A) root weight and (B) stomatal conductance on Cherries ‘Regina’loam soil, 15 L/ha applied four times (60 L per season). Treatment means differed significantly based on Duncan’s Multiple Range Test at the 5% significance level (p ≤ 0.05).
Similar trends were observed in walnut ‘Chandler’, where pruning root weight increased from 52 g to 68 g following application, demonstrating substantially greater root development (Figure 4).

Figure 4: Physiological effects after Release LPH™ application on pruning root weight and (B) stomatal conductance on Walnuts ‘Chandler’ applied at 15 L/ha applied four times (60 L per season). *Treatment means differed significantly based on Duncan’s Multiple Range Test at the 5% significance level (p ≤ 0.05).
These improvements extended to yield performance and fruit quality. In ‘Autumn Crisp’ table grapes, RELEASE LPH™ increased berry diameter, cluster weight, clusters per plant and fruit firmness relative to untreated controls (Table 1).

Table 1. Fruit quality parameters of ‘Autumn Crisp’ after 15 L/ha Release LPH™ applied four times (60 L per season). Treatment means differed significantly based on Duncan’s Multiple Range Test at the 5% significance level (p ≤ 0.05).
Fruit size distribution analyses further showed a greater proportion of larger, commercially desirable fruit classes in both walnut and cherry crops following treatment (Figures 6 and 7), indicating improved pack-out potential and marketable yield.
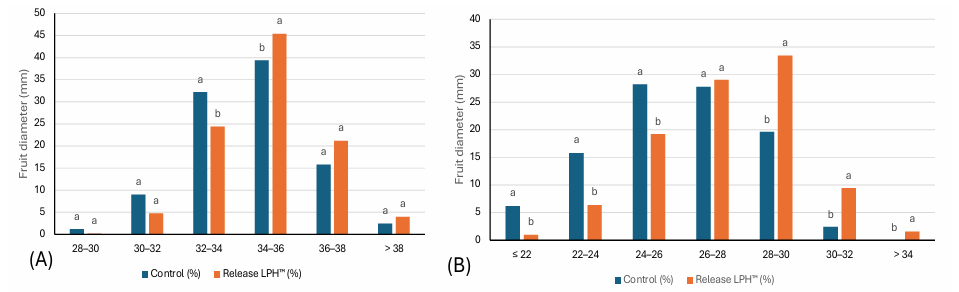
Figure 6: Fruit size of (A) Walnut ‘Chandler’ and (B) Cherries ‘Regina’ after 15 L/ha RELEASE LPH™ applied four times (60 L per season). Treatment means differed significantly based on Duncan’s Multiple Range Test at the 5% significance level (p ≤ 0.05).

Figure 7: Fruit size of distribution of Walnut ‘Chandler’ after 15 L/ha RELEASE
LPH™ applied four times (60 L per season). Treatment means differed significantly based on Duncan’s Multiple Range Test at the 5% significance level (p ≤ 0.05).
Conclusion
RELEASE LPH™ functions as a soil prebiotic, stimulating indigenous microbial activity rather than introducing external organisms. By enhancing microbial nutrient cycling and promoting root development through seaweed-derived bioactives, it strengthens rhizosphere function and root–shoot signalling, improving nutrient uptake, root performance and yield potential.
The field trials and performance data were generated in collaboration with Anasac, a Chilean multi-national agribusiness founded in 1948 that develops and supplies crop protection, plant nutrition and biostimulant technologies across Latin America. Anasac’s technical teams designed and managed the multi-environment trials, whose rigorous methodology, data collection and independent evaluation underpin the results presented. We gratefully acknowledge their partnership in validating the agronomic and biological performance of RELEASE LPH™ under commercial conditions.
Contact an Agri Technovation crop specialist or the Laeveld Agrochem agent in your area for specialised crop and application advice.
References:
1. Bashan, Y., de-Bashan, L.E., Prabhu, S.R., & Hernandez, J.P. (2014). Advances in plant growth-promoting bacterial inoculant technology: formulations and practical perspectives. Plant and Soil, 378, 1–33.
2. Canarini, A., Kaiser, C., Merchant, A., Richter, A., & Wanek, W. (2019). Root exudation of primary metabolites: mechanisms and ecological roles. New Phytologist, 221, 704–718.
3. Craigie, J.S. (2011). Seaweed extract stimuli in plant science and agriculture. Journal of Applied Phycology, 23, 371–393.
4. Kaminsky, L.M., Trexler, R.V., Malik, R.J., Hockett, K.L., & Bell, T.H. (2019). The inherent instability of seed-introduced microbial inoculants in agricultural soils. Environmental Microbiology, 21, 164–175.
5. Khan, W., Rayirath, U.P., Subramanian, S., et al. (2009). Seaweed extracts as biostimulants of plant growth and development. Journal of Plant Growth Regulation, 28, 386–399.
6. Liu, H., Brettell, L.E., Qiu, Z., & Singh, B.K. (2020). Microbiome-mediated stress resistance in plants. Trends in Plant Science, 25, 733–743.
7. Nardi, S., Pizzeghello, D., Muscolo, A., & Vianello, A. (2002). Physiological effects of humic substances on higher plants. Soil Biology & Biochemistry, 34, 1527–1536.
8. Sakakibara, H. (2006). Cytokinins: activity, biosynthesis, and translocation. Annual Review of Plant Biology, 57, 431–449.





